NanoComp researchers: Dr Samira Kaissi, Commercial lead, Dr Ajay Kumar, Technical Lead Scientist and Dr Promita Bhattacharjee, Biology Lead Scientist.
NanoComp Advisors: Prof. Kieran Hodnett, Project Contributor/Advisor, Prof. Ake Rasmusen, Project Contributor/Advisor, Co-inventor, Dr Peter Davern, Project Contributor/Advisor, University of Limerick.
Challenge
Current trends in small molecule drug substance development show a high prevalence of substances that fit into Classes II & IV in the Biopharmaceutics Classification System and as such have an inherent low solubility in-vivo, Figure below. While these compounds are highly efficacious in treating the underlying disease of their intended patient populations, they require enhanced formulation strategies to develop a viable drug product therapy that is effective when administered to the same patient population.
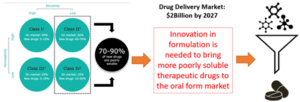
For several of these novel compounds, no viable formulation strategy or manufacturing process can overcome the inherent low solubility of the molecule, resulting in the potential therapy being discarded. For others, a challenging, labour intensive undertaking is required, that calls for highly customized excipients and complex manufacturing processes, to allow for the development of a viable drug product. These manufacturing processes include wet milling and/or traditional, nano, or supercritical assisted spray drying, all of which are utility and resource heavy unit operations when run at scale in drug product manufacturing. The result of this current state-of-art is a supply system that has high cost of goods and capital, is labour and equipment intensive, vulnerable towards single points of failure and embeds challenges around its long term economic and environmental sustainability. In turn, this does however create a strong unmet need for innovation to create new technologies that are more economical and sustainable alternatives, which deliver comparable or better final drug products.
Solution
The NanoComp technology is novel and has been developed to provide a simple and elegant solution that provides a comparable or better product which benefits the end users. In conjunction with this benefit, the innovation is forward facing by also actively improving the sustainability, efficiency, and throughput potential of the oral drug product manufacturing process.
NanoComp, a patented oral drug formulation platform, utilizes simple and efficient manufacturing processes to change insoluble APIs into powder that is ready for tableting while adding superior dissolution and stability to the final drug product form. The technology has been developed by Prof. Sarah Hudson and her team at the University of Limerick as part of her on-going research within SSPC’s Materials. The technology is currently undergoing commercial evaluation in conjunction with Enterprise Ireland for pharmaceutical small molecule drug development.
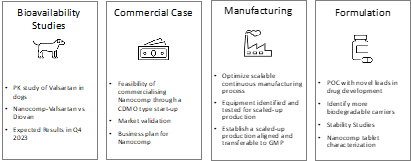
The result of the current state-of-art for improving water solubility of BCS class II and IV APIs is a supply system that has high cost of goods and capital, is labour and equipment intensive, vulnerable towards single points of failure and embeds challenges around its long term economic and environmental sustainability. In turn, this does however create a strong unmet need for innovation to create new technologies that are more economical and sustainable alternatives which deliver comparable or better final drug products. The pathway towards establishing the commercial potential of this technology is composed of four key challenges; demonstration of bioavailability, establishment of the commercial case, and activities to further develop the manufacturing and formulation aspects of the technology, detailed below.
Impact
In terms of societal impact, the NanoComp technology is seeking to provide two key impacts as an alternative manufacturing platform technology. The first of these is a viable pathway for achieving greater input efficiencies around the provision of reliable, suitable, and affordable medicines, which in turn, gives companies an option through which they can create greater access to existing medicines or create medicines capable of treating unmet needs within the population. The second impact is around the drive towards future smart technologies that are inherently sustainable through lower utility, materials, and water treatment demands, which in turn ensure a lower environmental impact footprint.
















